Product Description
The O2 OASYS Oral / Nasal Airway System is cleared as a 510 K Medical Device by the ENT and Dental Divisions of the FDA:
- Mandibular Repositioner for treatment of Snoring and Sleep Apnea;
- Nasal Dilator for improved breathing through the nose.
It also treats Sleep Disordered Breathing by addressing Upper Airway Resistance in the nasal region. The Lingual Tongue Button component is optional, but adds tongue training and repositioning for improved tongue placement, better swallowing and improved sleep treatment results even in Severe OSA patients.
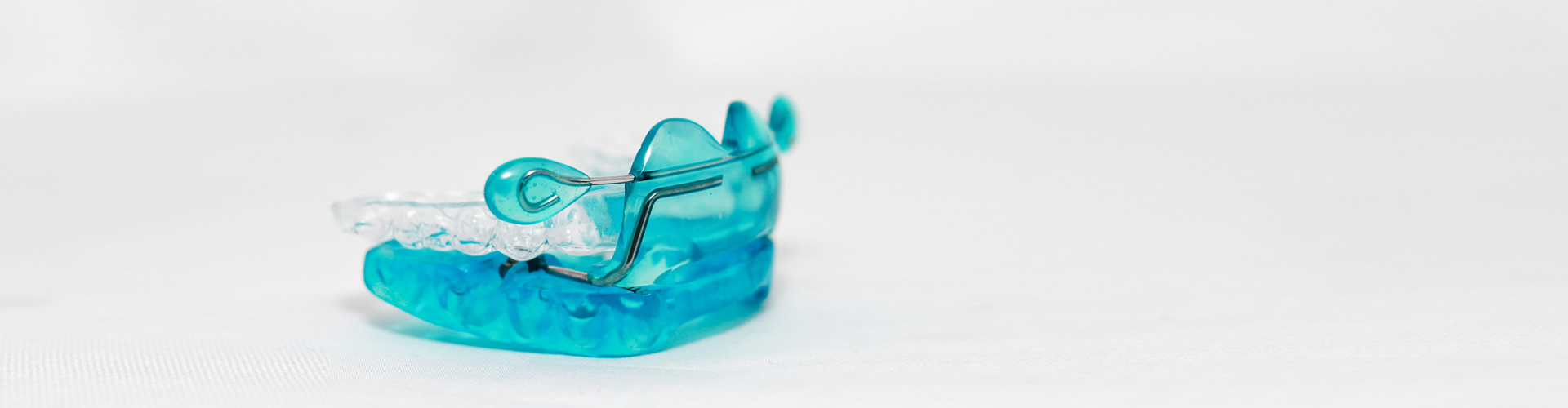
The Anterior Labial Shield adjusts easily forward and backward, 8-15mm on parallel bars, with an easy locking system and a MM Scale. A Clear Splint acts as a cushion to reduce pressure on the upper anterior teeth and being unattached, provides greater freedom of movement.
The Upper Oasys splint can be a full coverage or open lingually from #7-10, to encourage tongue placement at the incisal papilla. Patented Nasal Dilators increase airflow through the nose with internal extensions in the maxillary vestibule that stretch the nasiolabial fold, easily adjusted intraorally.
Patented Lingual Tongue Buttons extend off the lower splint in the posterior molar region to guide the tongue up and forward to the palate. This also makes the patient’s swallowing more dynamic, establishing a better tongue posture. The OASYS is a good choice for a patient wearing a good fitting Upper Denture, which will be worn in place of the upper splint.
The OASYS can also be used with Invisalign. The Upper Invisalign tray is worn in place of the Upper Oasys splint and the lower splint is designed to function over the Invisilign trays and adjusted during treatment.